Was die Raman-Spektroskopie Ihnen sagen kann
Die Raman-Spektroskopie kann Informationen über die Schwingungsmoden (d. h. bestimmte, zeitlich stationäre Eigenschaften einer Welle) eines Moleküls oder Kristalls liefern. Wir können Raman-Spektren für die chemische und strukturelle Charakterisierung verwenden.
Sie können Raman-Spektren interpretieren, um Chemikalien zu identifizieren und Informationen über ihre Struktur zu erhalten. Die Raman-Streuung entsteht aus der Wechselwirkung zwischen Licht und molekularen Schwingungen. Diese Schwingungen reagieren sehr empfindlich auf chemische und strukturelle Änderungen und ermöglichen Ihnen die Erkennung feinster Unterschiede in der molekularen Umgebung. Mit Ausnahme von reinen Metallen erzeugen generell alle Stoffe Raman-Spektren.
Was ist ein Raman-Spektrum?
Die Ergebnisse unserer Messungen mittels Raman-Spektroskopie stellen wir grafisch in Form von Raman-Spektren dar. Die Y-Achse stellt die Intensität des gestreuten Lichts dar, während die X-Achse die Lichtenergie (-frequenz) abbildet. Da wir uns für die Frequenzverschiebung der Raman-Streuung interessieren, stellen wir die Form der von der X-Achse abgebildeten Frequenzen im Verhältnis zu denen des Lasers dar. Wir bezeichnen die X-Achse als Raman-Verschiebung (dargestellt durch die Einheit cm-1).
Welche Informationen kann mir die Raman-Spektrokopie liefern?
Kurz gesagt, nutzen wir die Merkmale des Raman-Spektrums wie folgt:
- Die Raman-Verschiebungen und entsprechenden Intensitäten aller Raman-Banden der Probe
Wir können die Probe anhand des spektralen Fingerabdrucks identifizieren. - Veränderungen der Raman-Spektren bei unterschiedlicher Ausrichtung oder Polarisation
Die Intensitäten und Positionen der Raman-Banden können sich mit der jeweiligen Ausrichtung der Probe ändern. Wir können dies sichtbar machen, indem wir die Polarisation des Anregungslasers und des gesammelten Raman-Lichtes drehen. Die polarisierte Raman-Spektroskopie liefert Informationen zur Symmetrie und Ausrichtung anisotroper Materialien.
- Veränderungen einzelner Banden
Eine Raman-Bande kann sich verschieben (Position), verengen oder verbreitern (Breite) oder in der Intensität (Höhe) variieren. Diese Veränderungen können Druck-/Zugspannungen in der Probe, Veränderungen der Kristallinität bzw. der Materialmenge sichtbar machen. - Veränderungen im Spektrum an verschiedenen Positionen auf der Probe
Diese können Ungleichmäßigkeiten (Heterogenität) im Material sichtbar machen. Sie können einzelne Punkte auswerten oder systematisch eine Reihe von Punkten messen (was Raman-Bilddaten mit Darstellung der Zusammensetzung, Spannung, Kristallinität usw. ermöglicht)
Ein Raman-Spektrum besteht daher aus verschiedenen Banden, die jeweils einer Schwingungsmode zugeordnet werden können. Jedes Material besitzt ein einzigartiges Spektrum und kann anhand dessen identifiziert werden. Einige Forscher versuchen, jede einzelne Raman-Bande und ihre Beziehung zu den Schwingungsmoden vollständig zu verstehen. Die meisten Analytiker identifizieren die Proben jedoch einfach anhand einer Spektrenbibliothek.
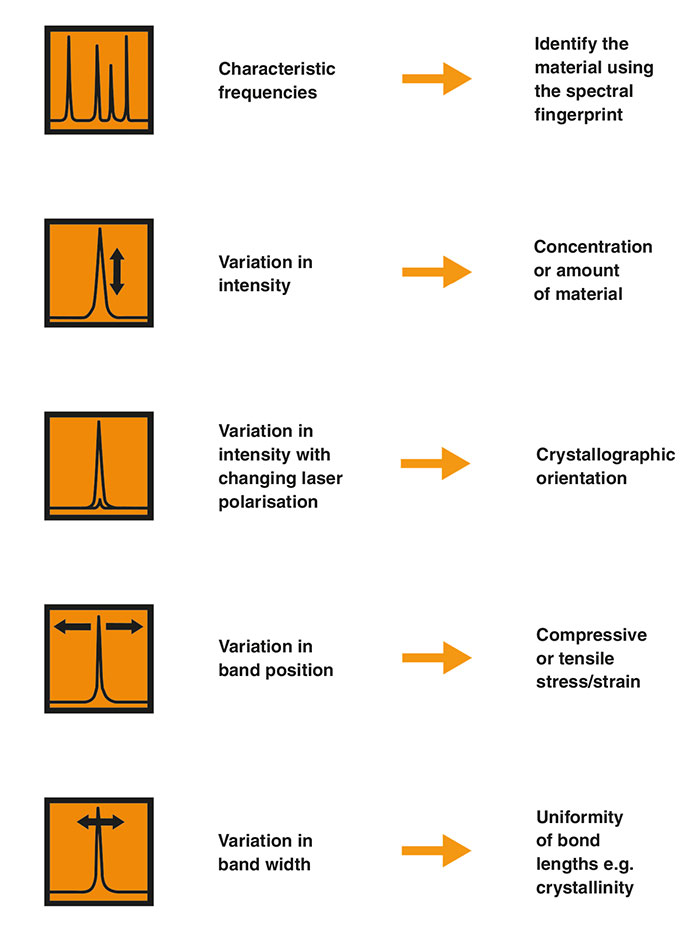
Merkmale eines Raman-Spektrums
Auswertung des Raman-Spektrums
Eine Möglichkeit, ein Raman-Spektrum zu verstehen, besteht darin, die molekularen Funktionsgruppen als separate Einheiten zu betrachten. Dies erleichtert die Interpretation des Raman-Spektrums von Kristallen mit einer regelmäßigen Anordnung identischer Atome, die alle gleich konfiguriert sind. Die Kohlenstoffatome in einem Diamanten sind beispielsweise in einer regelmäßigen tetraedrischen Gitterstruktur gebunden. In diesen Fällen ist oft nur eine dominante Raman-Bande zu erkennen, da der Kristall nur eine molekulare Umgebung besitzt.
Polystyrol dagegen besitzt ein komplexeres Raman-Spektrum. Das Molekül ist weniger symmetrisch und enthält neben den Kohlenstoffatomen auch Wasserstoffatome. Außerdem sind die Bindungen zwischen den Atomen unterschiedlicher Art.
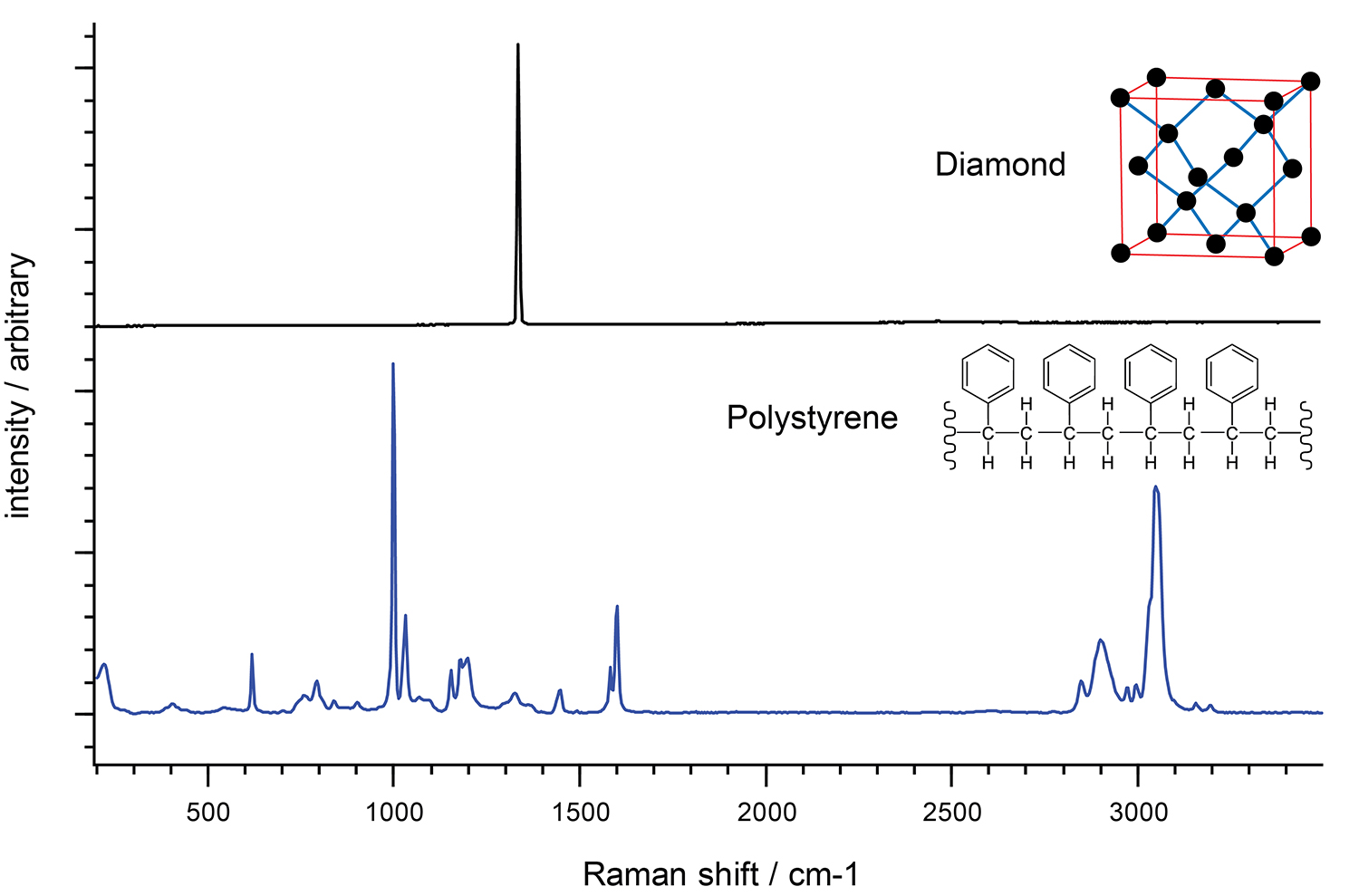
Raman-Spektrum von Diamant und Polystyrol. Das Raman-Spektrum von Polystrol ist aufgrund der unterschiedlichen Bindungsarten komplexer als das von Diamant.
Charakteristische Schwingungsfrequenzen chemischer Bindungen
Die Schwingungsfrequenzen hängen von den Massen der beteiligten Atome und der Stärke der Bindungen zwischen ihnen ab. Bei schweren Atomen und schwache Bindungen ist die Raman-Verschiebung gering. Bei leichten Atomen und starken Bindungen ist die Raman-Verschiebung hoch.
Im Polystyrol-Spektrum sehen wir die hochfrequenten Kohlenstoff-Wasserstoff (C-H)-Schwingungen bei etwa 3.000 cm-1. Die niederfrequenten Kohlenstoff-Kohlenstoff (C-C)-Schwingungen betragen etwa 800 cm-1. Die C-H-Schwingungen haben eine höhere Frequenz als die C-C-Schwingungen, da Wasserstoff leichter ist als Kohlenstoff.
Ähnlich verhält es sich mit den Schwingungen zweier Kohlenstoffatome, die durch starke Doppelbindungen (C=C) bei etwa 1.600 cm-1verbunden sind. Diese Frequenz ist höher als bei zwei Kohlenstoffatomen, die eine schwächere Einfachbindung eingehen (C-C, 800 cm-1).
Anhand dieser einfachen Regeln lassen sich viele Merkmale der Raman-Spektren erklären.
Raman-Verschiebungen reagieren empfindlich auf benachbarte Bindungen
Bei genauer Betrachtung der Raman-Spektren lassen sich noch feinere Effekte erkennen. Beispielsweise treten die C-H-Schwingungen von Polystyrol in zwei Banden auf, bei etwa 2.900 cm-1und 3.050 cm-1. Die Kohlenstoffe in der ersten Bande sind Bestandteil aliphatischer Kohlenstoffketten, die Kohlenstoffe in der zweiten Bande dagegen sind Bestandteil aromatischer Kohlenstoffringe.
Die Schwingungen eines komplexen Moleküls können so betrachtet werden, als würden sie teilweise aus vielen einfachen zweiatomigen Schwingungen bestehen. Für ein vollständiges Verständnis des Raman-Spektrums sollten jedoch auch die Schwingungen größerer Atomgruppen berücksichtigt werden. Das Raman-Spektrum von Polystyrol hat beispielsweise eine Bande bei 1.000 cm-1. Dies liegt an der sich ausdehnenden und zusammenziehenden „Atmungsmode“ des aromatischen Kohlenstoffrings im Polystyrol.
Niederfrequente Raman-Banden
Sie können auch Schwingungs- und Rotationsmoden von Molekülen mit niederfrequenten Raman-Verschiebungen (unter 100 cm-1)untersuchen. Diese entstehen durch sehr schwere Atome oder durch sehr großflächige Schwingungen, wie beispielsweise eine Schwingung des gesamten Kristallgitters. Mit den Raman-Instrumenten von Renishaw können Sie diese Moden untersuchen. Sie können ein breites Spektrum an Materialien und Kristallen erforschen und dabei leicht zwischen verschiedenen kristallinen Formen (Polymorphen) und Schichtstrukturen unterscheiden.
Wie können Stoffe mittels Raman-Spektroskopie identifiziert werden?
Wir können unbekannte Materialien anhand ihres einzigartigen spektralen Raman-Fingerabdrucks („Fingerpint“) identifizieren, in der Regel mithilfe von Software-Suchläufen in Spektrenbibliotheken. Wir verwenden Raman-Banden im Fingerprint-Bereich (von 300 cm-1bis 1.900 cm-1), um Moleküle zu identifizieren.
Idealerweise verwenden Sie ein Raman-Gerät mit hoher spektraler Auflösung über den gesamten Raman-Bereich. Dadurch erhalten Sie genaueren Einblick in die chemische Spezifität und können eine breitere Auswahl an Materialien identifizieren, differenzieren und untersuchen.
Wissenswertes über Raman-Spektroskopie
Neu auf dem Gebiet der Raman-Spektroskopie? Machen Sie sich schnell mit den Grundlagen der Raman-Analyse vertraut.
Mit Raman-Spektroskopie Kristallinität und Polymorphismus verstehen
Durch den Vergleich der Raman-Spektren von Materialien können Sie Unterschiede in ihrer Struktur untersuchen. Sie könnten den Grad der Kristallinität quantifizieren und ähnliche Kristallformen (Polymorphismus) derselben Chemikalie unterscheiden. Dazu würden Sie ein Raman-Spektrometer mit hoher spektraler Auflösung benötigen wie beispielsweise das konfokale Raman-Mikroskop inVia™.

Raman-Spektren zweier Polyethylen-Proben, die unterschiedliche Intensitäten und Bandbreiten zeigen. Diese spektralen Abweichungen sind auf unterschiedliche Kristallinitätsgrade zurückzuführen.
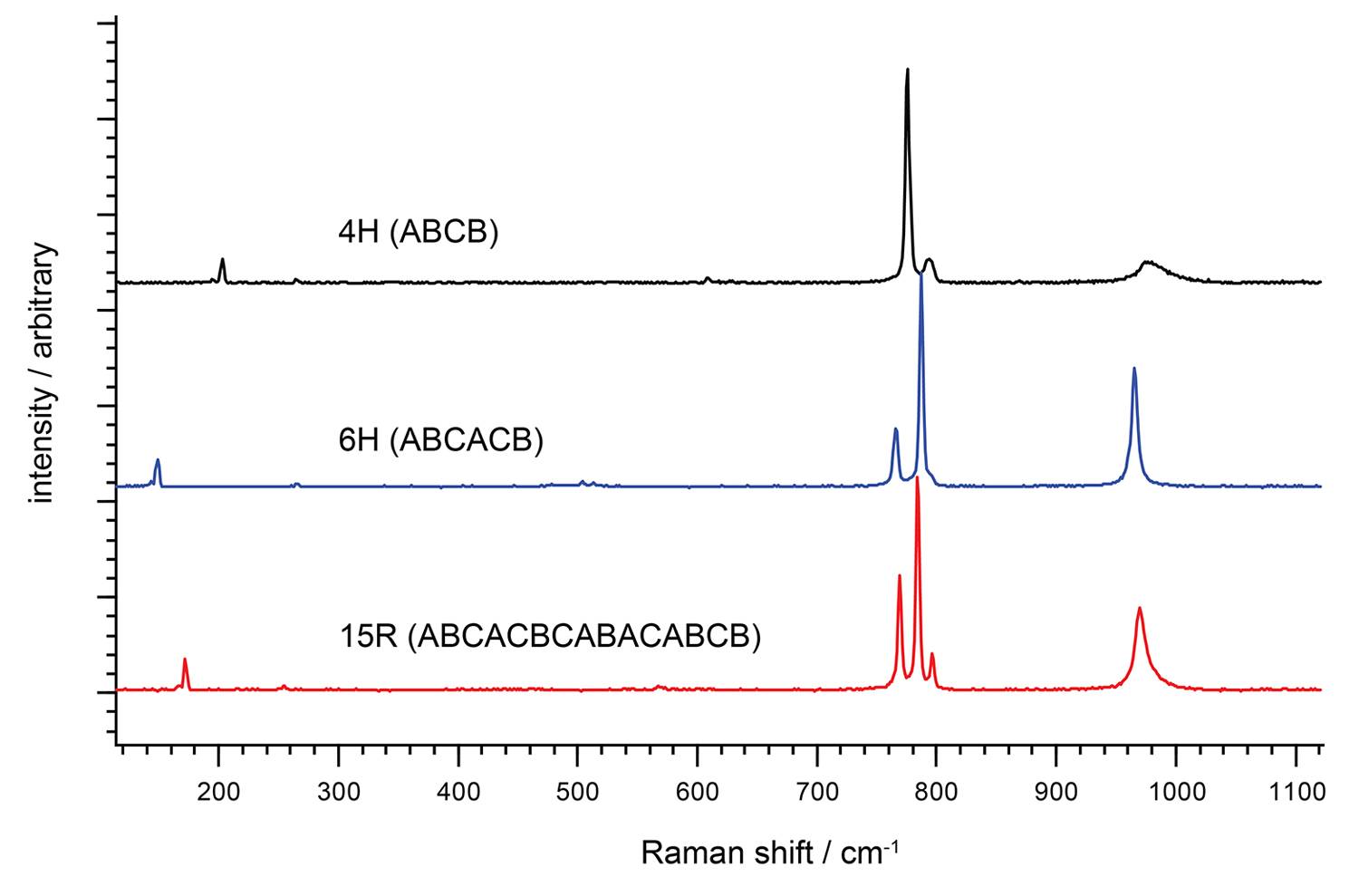
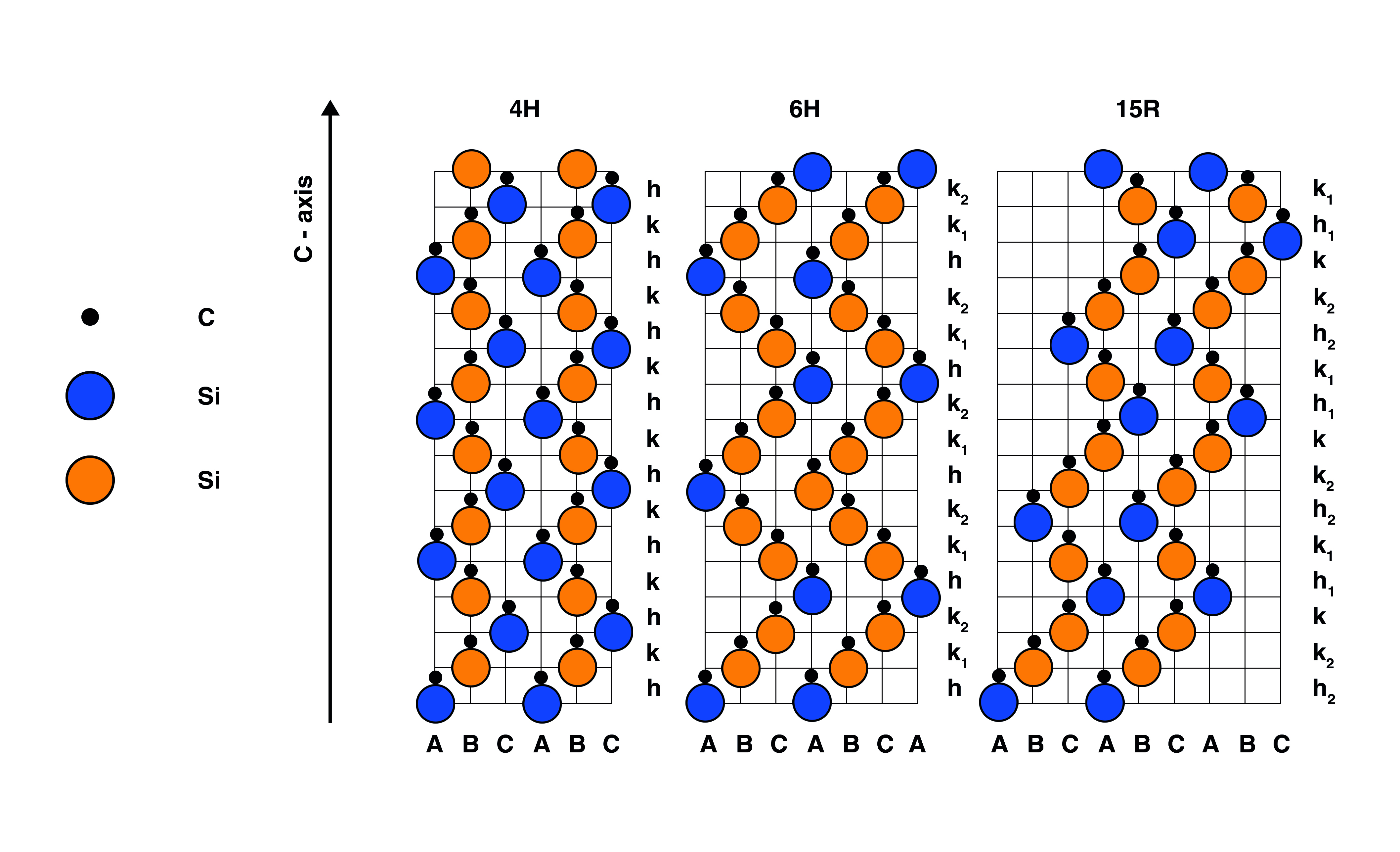
Links: Spektren von drei verschiedenen polymorphen Strukturen von Siliciumcarbid (SiC), einem wichtigen Halbleitermaterial.
Rechts: Kristallstruktur von SiC-Polytypen: 4H-SiC; 6H-SiC und 15R-SiC.
Die Polytypen 4H, 6H und 15R von SiC können leicht mittels Raman-Spektroskopie unterschieden werden. Die kontrollierte Herstellung von SiC-Polytypen ist wichtig für industrielle Halbleiteranwendungen.Was ist Raman-Spektroskopie?
Erfahren Sie noch mehr über die Raman- und Photolumineszenz- (PL-)Spektroskopie. Wir beantworten Ihre Fragen zur Raman-Mikroskopie, schnellen Raman-Bildgebung, Datenauswertung, Fluoreszenz und zu begleitenden Analyseverfahren.
Wissenswertes über Raman-Spektroskopie